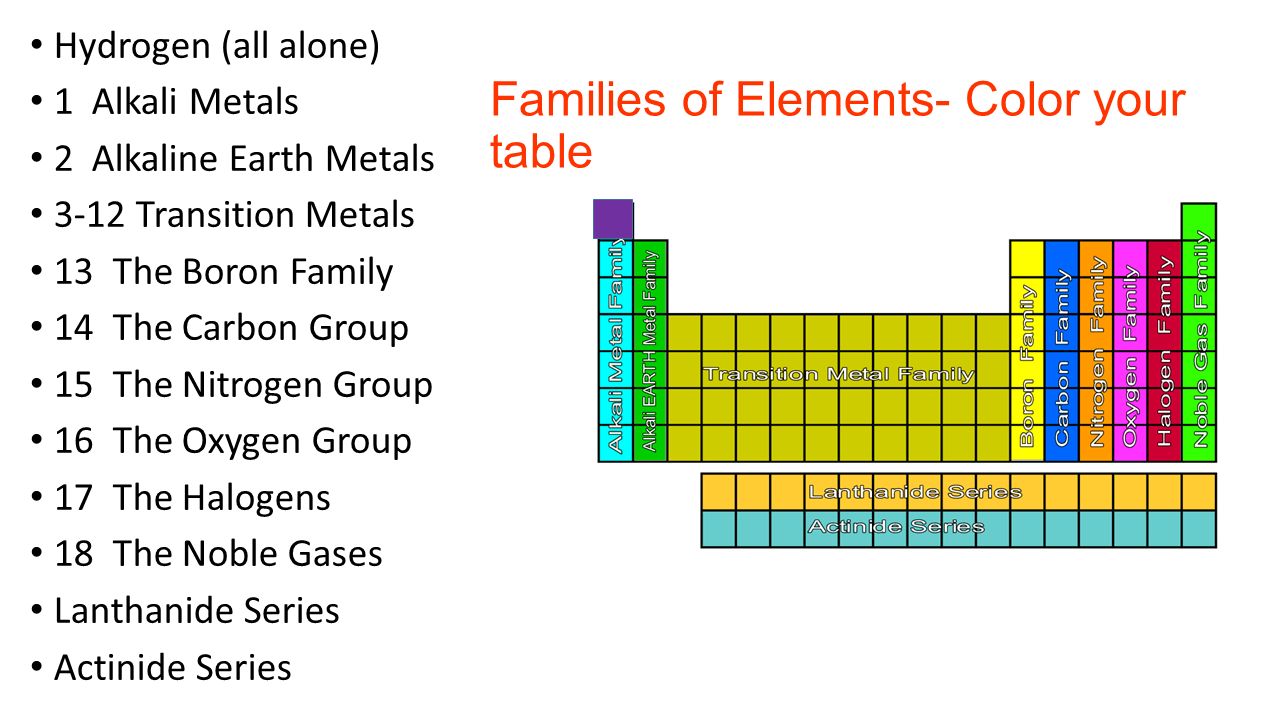
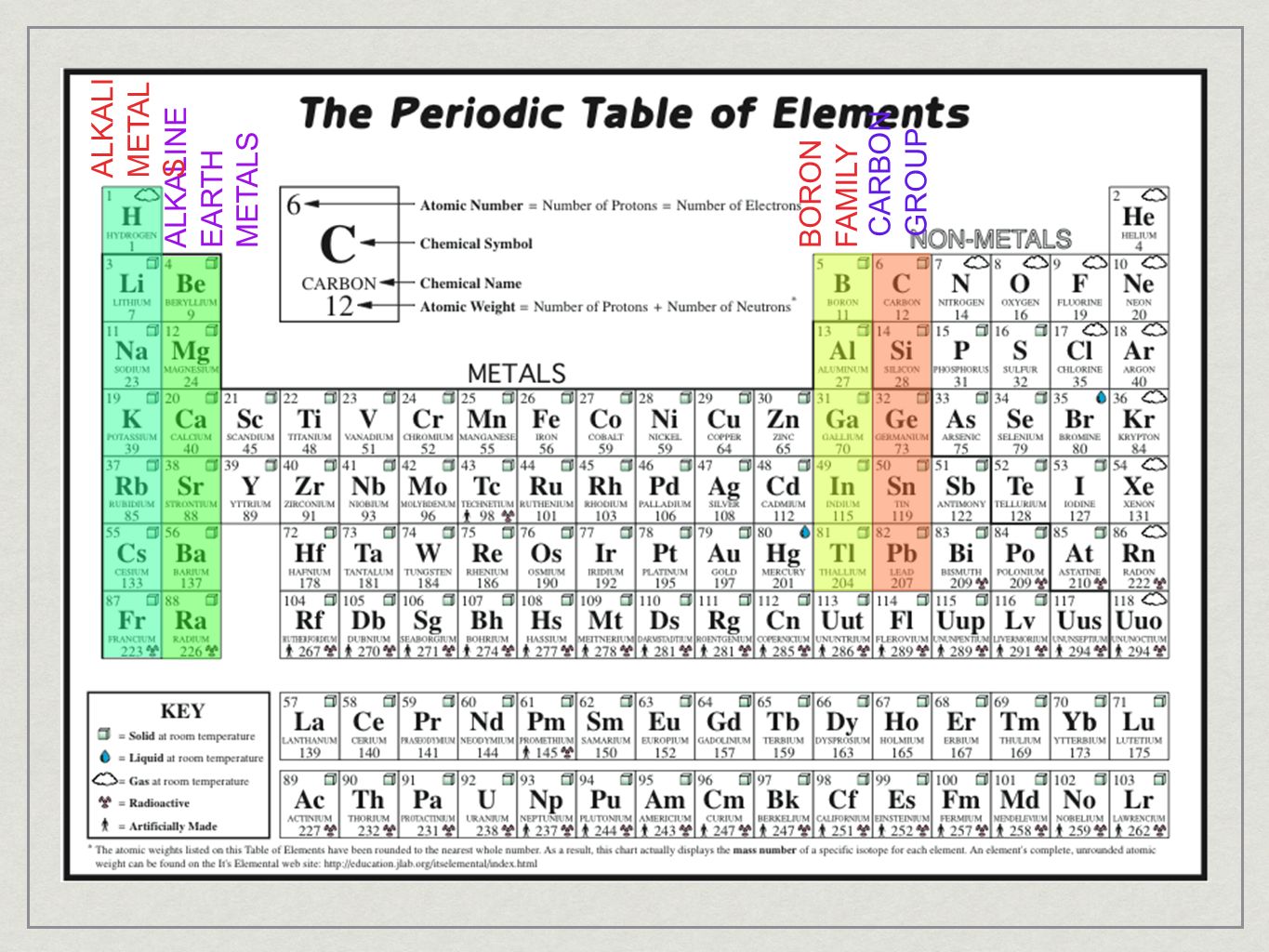
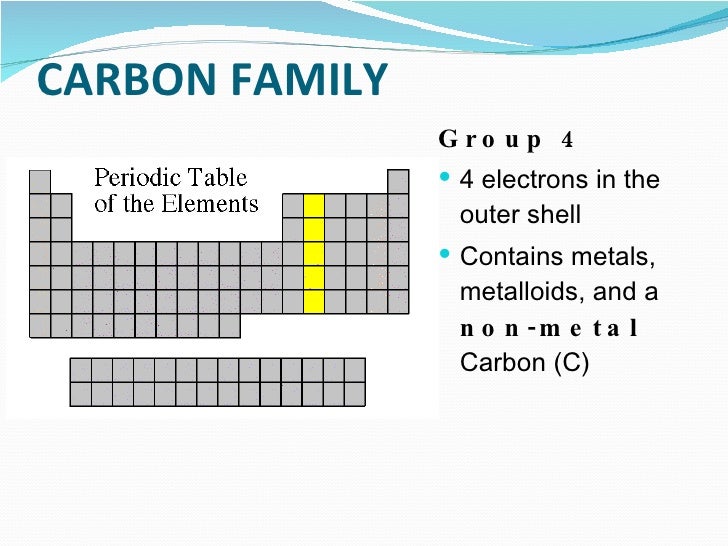
Glossary. Group A vertical column in the periodic table. Members of a group typically have similar properties and electron configurations in their outer shell.
A group in the Periodic Table is any column of elements with common properties denoted by the electrons in the outermost layer. There are …
A group or family is a vertical column in the periodic table. Groups usually have more significant periodic trends than periods and blocks, explained below. Modern quantum mechanical theories of atomic structure explain group trends by proposing that elements within the same group generally have the same electron configurations in their valence
Interactive periodic table with dynamic layouts showing names, electrons, oxidation, trend visualization, orbitals, isotopes, and compound …
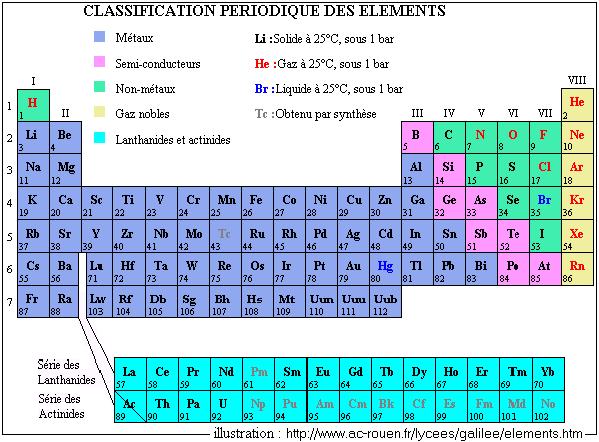
An up-to-date periodic table with detailed but easy to understand information
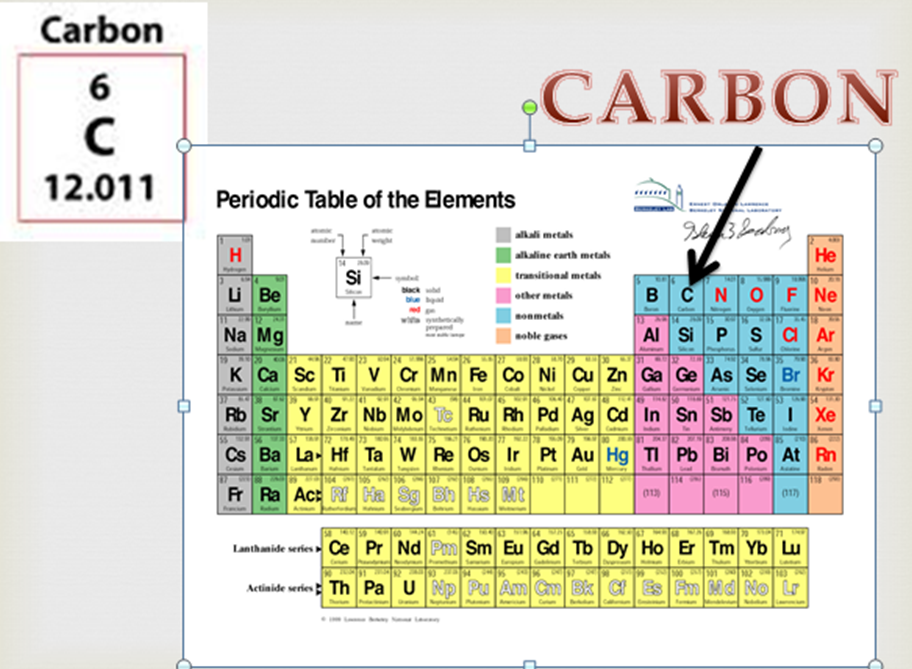
Photographs and descriptions of many samples of the element Carbon in the Periodic Table.
In chemistry, a group (also known as a family [citation needed]) is a column of elements in the periodic table of the chemical elements.There are 18 numbered groups in the periodic table, and the f-block columns (between groups 3 and 4) are not numbered.


John Newlands table of the Elements first published February 7, 1863 Showing comparison to Modern table with group and period classification Note: only H, Li, and Mg in correct placement both period and group
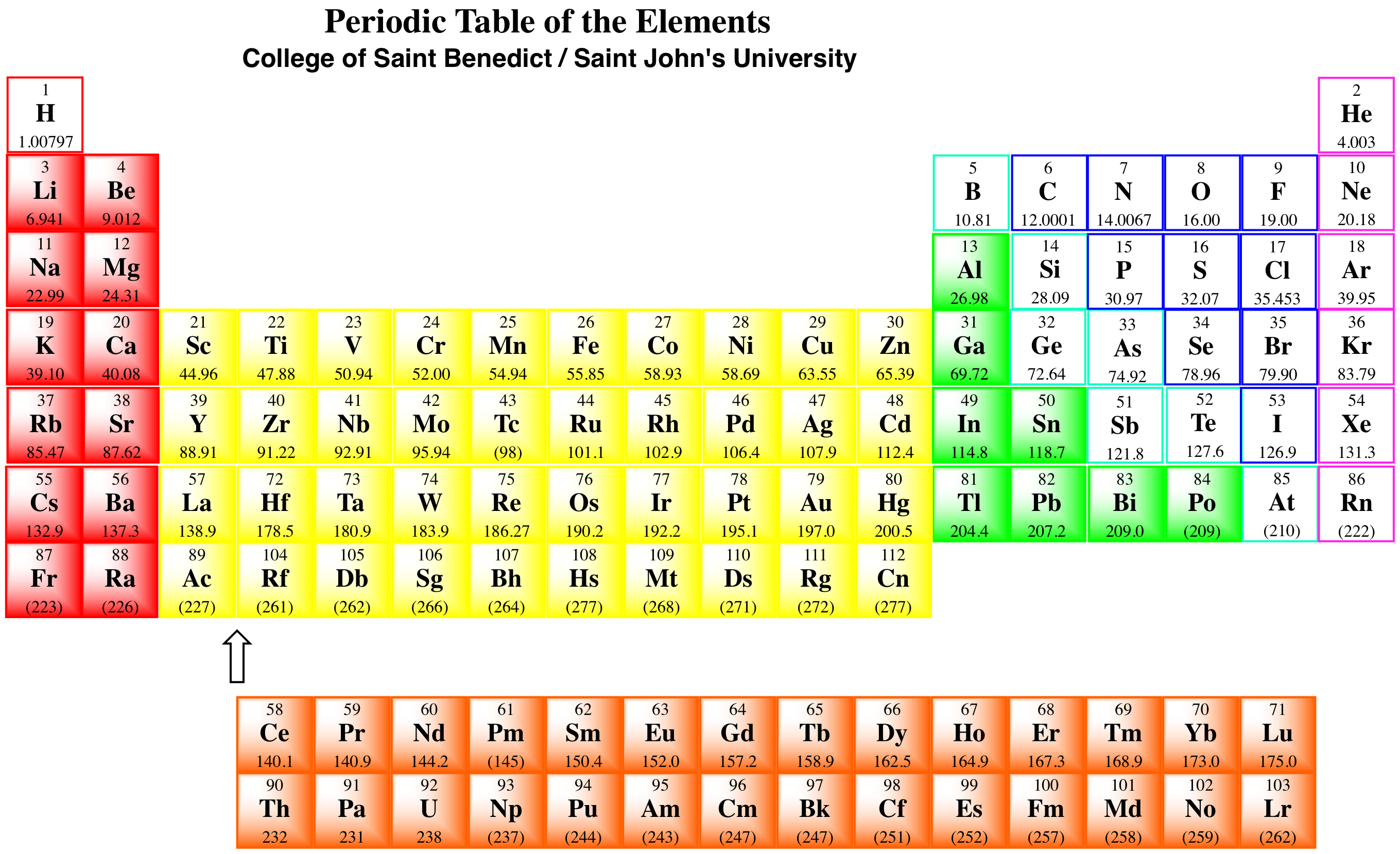
Periodic table of elements with information on chemical elements like gold, silver, iron, neon, helium, chlorine, copper, cobalt, and more

Explore the chemical elements through this periodic table
